The main interest of our lab is to understand the molecular mechanisms by which sensory neurons detect noxious stimuli, so-called nociceptors and how the properties of these neurons change from health to disease. We also work on all aspects of naked mole-rat biology (scroll past the pain section for more info.!)
Nociception versus pain
The International Association for the Study of Pain defines nociception as “the neural process of encoding noxious stimuli”, whereas pain is defined as being more than just the initial sensation of the noxious stimulus, “An unpleasant sensory and emotional experience associated with, or resembling that associated with, actual or potential tissue damage.” Nociceptors express a range of ion channels and receptors that when activated can result in nociceptor activation. A classic example is the burning sensation evoked by chilli peppers, which is due to a chemical called capsaicin that activates an ion channel called TRPV1.
One important factor to consider in pain research is sex and how this may affect both pain and its treatment. Researchers have traditionally used male animal models, but the Smith Lab supports the need to consider use of males and females when designing a study. Here is a podcast Ewan did with The Naked Scientists about pain and sex, based upon a perspective piece in Nature from Prof. Jeffrey Mogil.
Here is a recent lecture that Ewan gave as part of the Cambridge Alumni Festival: Pathways to pain: how to cure, not cause!
Acid sensing
In humans, lemon juice and vinegar both cause a stinging sensation when splashed over a cut in the skin, which is due to their acidic nature. Acid is sensed by a variety of different sensory neuron receptors and the ability of acid to activate sensory neurons and evoke nocifensive behaviour is common to many species of the animal kingdom, as we have recently reviewed. Tissue acidosis occurs in a variety of different pathological conditions, including some inflammatory states and certain cancers. The Lab a long-standing interest in how the acid-sensing ion channel (ASIC) family function and their contribution towards pain. Work from our lab has identified critical domains involved in ASIC activation by acid and investigated species differences in ASIC function. We have also recently developed an interest in the function of proton-sensing G protein-coupled receptors (PS-GPCRs), one of which, GPR65 plays a key role in inflammatory pain, through its activation on fibroblast-like synoviocytes (FLS, a key cell in the knee joint infrastructure that secretes substances into the synovial fluid), which induces pro-inflammatory mediator release and neuronal sensitisation.
Neuronal excitability
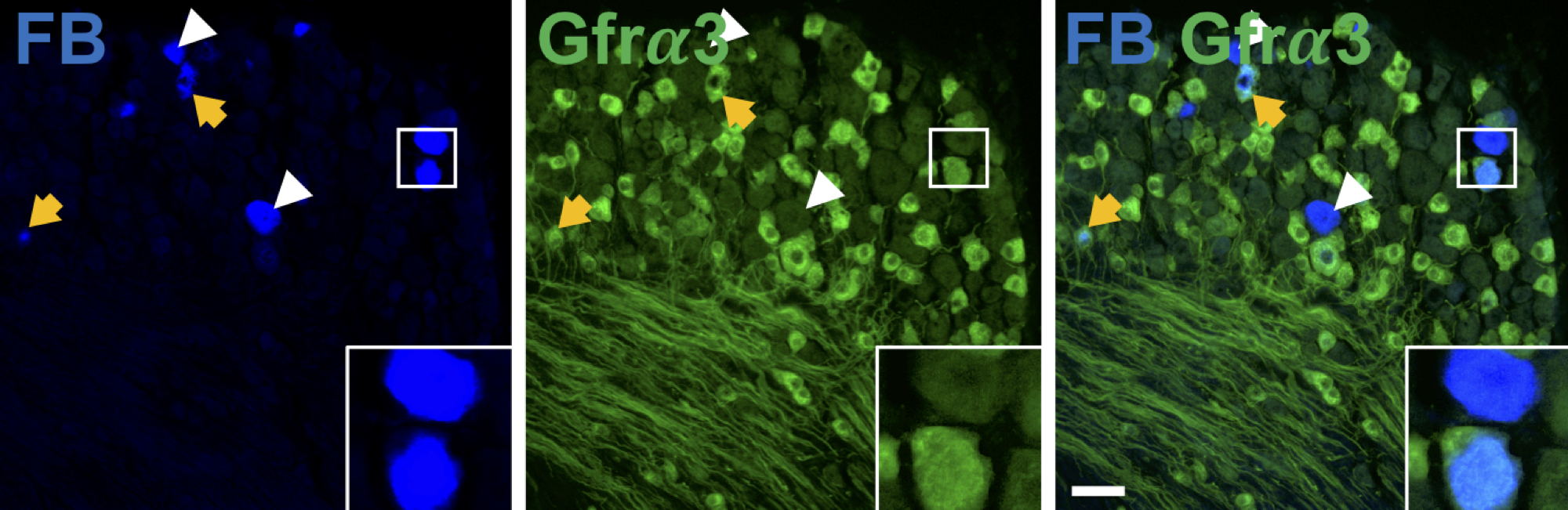
We are also interested in bridging the gap between understanding how sensory neurons are activated in the normal, uninjured state and how their function is altered in conditions that are associated with pain, in particular arthritis and inflammatory bowel disease. By understanding how sensory neurons are activated in pathological conditions, we hope to identify novel avenues for therapeutics. To study the properties of particular sensory neurons we use retrograde tracing techniques: a fluorescent substance is injected into the site of interest (e.g. knee joint, or distal colon) and then travels up the sensory nerve fibres to the cell bodies - we can then use the fluorescence to specifically characterise the function of neurons coming from the site of interest and their functionality changes in disease. For example, in the diagram below, the left image shows fast-blue (LB) labelled lumbosacral neurons, yellow arrows represent FB+ neurons showing expression of the marker of interest, in this case GDNF family receptor alpha-3 (Gfrα3, middle panel) and white arrowheads represent FB+ neurons that do not express the marker of interest. The right panel shows the merged image with the inset showing examples of FB+/Gfrα3+ and FB+/Gfrα3- neurons. Scale bar 50 μm.
In 2019, we published a study using RNA-sequencing to identify 7 subtypes of colonic sensory neurons, which we then validated using scqRT-PCR, immunohistochemistry and Ca2+-imaging - future work will investigate the roles of these sensory neuron subtypes in health and disease. In a collaboration with David Bulmer, Nicolas Cenac and David Hughes, we further demonstrated that the lipid metabolite 5-oxoETE is found at higher levels in constipation predominant irritable bowel syndrome and that this drives pain by activating one of the seven colonic sensory neuron subtypes. You can explore gene expression and co-expression in the 7 colonic sensory subtypes here.
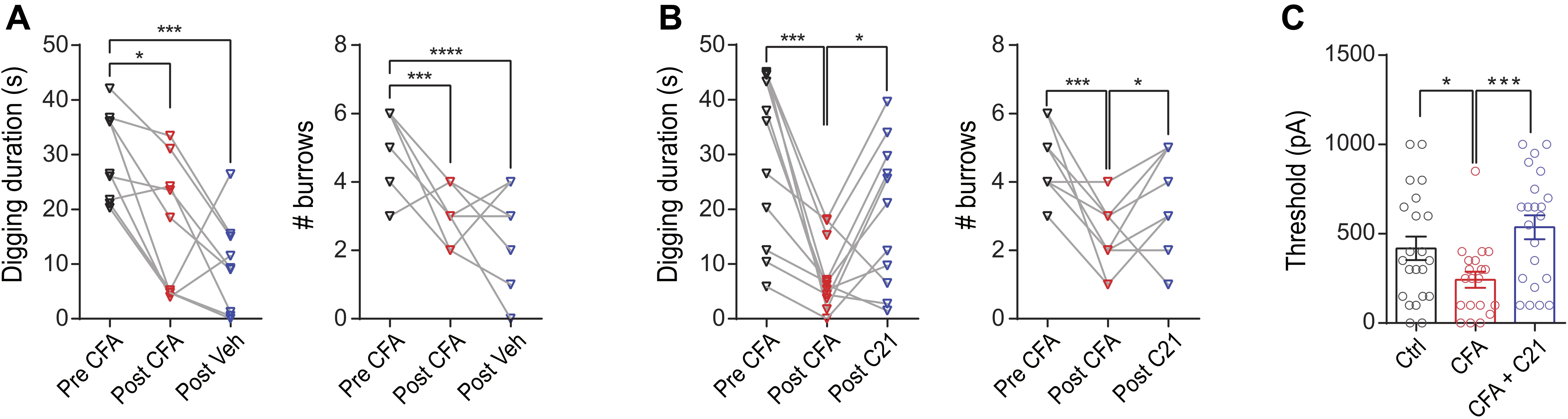
We also have also used retrograde tracing to show that TRPV1 expression increases in knee-innervating sensory neurons in mice during inflammation and that the inflammation-induced decrease in mouse digging behaviour (a measure of how pain affects spontaneous behaviour) is reversed by administration of a TRPV1 antagonist as shown in the figure below (A, experimental timeline; CFA = complete Freund's adjuvant to induce inflammation, B and C, 24-hours after CFA knee injection mice dig for less time and dig fewer burrows, which is reversed by administration of a TRPV1 antagonist, D and E, the antagonist has no effect on the digging behaviour or mice injected with saline). A lay summary of this work is provided here.
We have also recently begun to work with human synovial fluid to try to identify substances present in synovial fluid that may drive pain. Our initial findings demonstrate that synovial fluid from patients with osteoarthritis can sensitise mouse knee sensory neurons, whereas synovial fluid from healthy individuals does not, future work will investigate the mechanisms involved, but this method promises to accelerate bench-to-bedside treatments for arthritic conditions. A lay summary of this work can be read here. Alongside this, we have shown that FLS can be stimulated by pro-inflammatory factors to in turn release their own pro-inflammatory mediators and that co-culturing these stimulated FLS with knee-innervating sensory neurons causes neuronal excitability; a similar process can be induced by activating the PS-GPCR, GPR65.
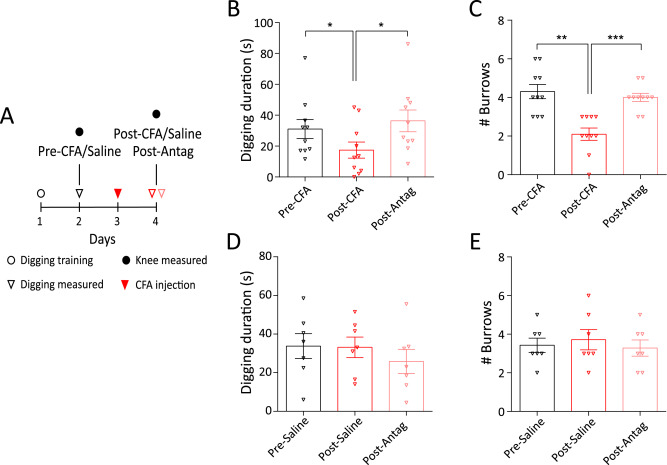
These results and those of others in the field demonstrate that targeting knee-innervating neurons may be a good way to alleviate joint pain and bypass the side effects associated with taking a drug systemically that acts on the whole body. To investigate this, in collaboration with Paul Heppenstall, we used AAV-PHP.S to deliver designer receptors exclusively activated by designer drugs (DREADD) constructs specifically to knee neurons. In a model of inflammatory joint pain using CFA, we found that whereas administration of vehicle (Veh, panel A below) had no effect on the CFA-induced decrease in digging behaviour, administration of the DREADD agonist compound 21 (C21, panel B below) reversed the CFA-induced decrease in digging behaviour and the hyperexcitability of knee-innervating neurons (panel C below), thus demonstrating potential for gene therapy in treating joint pain. You can read about this work here, which was highlighted in the journal Arthritis & Rheumatology.
An alternative method for modulating neuronal function is to induce expression of pharmacologically selective actuator modules (PSAMs), which are ion channel based methods for regulating excitability. In collaboration with Dave Bennett and Steve Middleton in Oxford we showed that activating an inhibitory PSAM relieved a variety of pain behaviours, which could be tied to inhibition of sensory neuron activity.
We have also begun to explore the function of mesenchymal stem cells (MSC) and MSC-derived extracellular vesicles (MSC-EV) in osteoarthritis. MSCs have been found to alleviate pain and improve joint function in osteoarthritis patients, but mechanisms for these effects are not fully understood. In a recent publication in Arhtiritis & Rheumatology, using a the destablisiation of the medial meniscus (DMM) mouse model of osteoarthritis, we demonstrated intra-articular administration of MSCs or MSC-EVs into the mouse knee joint, after the onset of pain behaviours, could reverse DMM-induced decrease in digging and improve quality of sleep without impacting joint degeneration. Moreover, knee neurons isolated from DMM mice were hyperexcitable, which was not observed in DMM mice treated with MSCs or MSC-EVs. Further work used nerve growth factor (NGF), a mediator associated with osteoarthritis pain, to sensitise sensory neurons in vitro and showed that MSC-EV application could reverse NGF-induced sensitisation. These results suggest that MSC-EVs are able to alleviate osteoarthritis pain through direct interaction with sensory neurons, results summarised in the cartoon below by Alice White, and work is on-going to discover through which MSC-EVs produce this analgesic effect.
Whereas traditional models of measuring pain have focused on seeing how an animal's response to a noxious stimulus changes under different conditions, we are keen to explore less interventional methods that may enable us to measure how on-going pain affects behaviour. Mice like digging and we have shown in studies mentioned above how inhibiting the activity of knee-innervating neurons can ameliorate reductions in digging behaviour observed in models of joint inflammation. We have recently expanded this to show that digging behaviour decreases in a mouse model of colitis, changes in digging correlating with gut histology. Moreover, we observed that whereas gabapentin and the non-steroidal anti-inflammatory drug (NSAID) meloxicam both reduced evoked pain at the site of injury, only meloxicam improved digging behaviour. This is likely because gabapentin has other influences on animal behaviour as noted by the fact that it also affects rotarod performance. Indeed, in the same study, in a survey of chronic pain patients, we found that individuals with osteoarthritis experienced better pain relief and fewer side effects with NSAIDs than with gabapentinoids, similar to results observed in mice. Similarly, in collaboration with Victoria Abraira and Ishmail Abdus-Saboor, using a technique to film freely behaving mice and analyse their modular usage of different behaviours (Motion Sequencing, MoSeq), we found that whereas gabapentin effectively ameliorated evoked knee pain in a mouse osteoarthritis model, it did not return the modular usage of behaviours to their basal state, suggesting that it induces side effects, which might affect its clinical efficacy. We therefore believe that including non-interventional assays, such as digging and MoSeq, alongside evoked pain assays will provide a more holistic assessment of animal behaviour and might improve translation from bed-to-bedside.
Naked mole-rats
The African naked mole-rat (Heterocephalus glaber) is a highly unusual mammal. Like certain ant, bee and termite species, naked mole-rats are eusocial, meaning that they live in large colonies with a sole, breeding female, the queen. Moreover, they are cold-blooded, live for over 35 years (similarly sized mice live for under a tenth of the time) are resistant to cancer and as others and we have shown, they have highly unusual nocifensive behaviours, in particular they do not find acid nocifensive. We identified that the molecular basis of the acid insensitivity displayed by naked mole-rats is due to an amino acid alteration in the voltage-gated sodium channel subunit NaV1.7: acid activates naked mole-rat acid sensors, but simultaneously blocks NaV1.7 to such an extent that action potentials are not generated. Interestingly, we find that neurons innervating the distal colon are activated by acid in the naked mole-rat (albeit less than in mouse), which can be explained by work looking at mice lacking NaV1.7 that exhibit normal visceral pain, i.e. NaV1.7 is more important for somatic than visceral pain.
We have also been involved in studies demonstrating the extreme hypoxia resistance of the naked mole-rat and have further shown how naked mole-rat cortical neurons are resistant to acid-induced cell death due to a decrease in the function of neuronal acid-sensing ion channels.
With regard to naked mole-rat cancer resistance. It was originally proposed that naked mole-rat cells were resistant to malignant transformation and thus providing a cell autonomous explanation for their cancer resistance. However, in a collaboration with Walid Khaled, we recently demonstrated that naked mole-rat cells can be transformed by oncogenes, just like the cells of mice, i.e. the cancer resistance of naked mole-rats is likely the result of something non-cell autonomous, perhaps an altered immune response...the search goes on! This research and the background to the project was covered in the Guardian. One recent collaborative investigation found that naked mole-rats have a slower somatic mutation rate, which might help to account for their lower incidence of cancer and healthy ageing, a brilliant Tweetorial of this was written by the lead author of the project Alex Cagan.
We now aim to conduct further comparative physiology and genetics with the naked mole-rat in order to identify molecules and circuits that can explain other aspects of their “odd” physiology, results from such work will lead to a greater understanding of how “normal” physiology works in other mammals including humans. You can find out more about people across Cambridge who are involved in research with the naked mole-rat, by going to the Naked Mole-Rat Initiative website. A recent comprehensive review of naked mole-rat biology can be read here.
You can read more about the naked mole-rat here, or, if you would prefer to watch some animations that we made, you can do so here