Transport proteins in normal and malignant cells
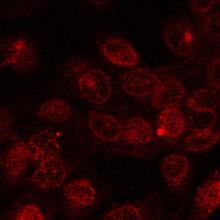
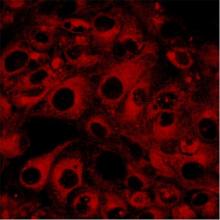
The ATP Binding Cassette superfamily of proteins contains several members that in mammalian cells are involved in drug transport. These include P-glycoprotein, multidrug-resistance associated proteins [1] and Breast Cancer Resistance Protein (BCRP). These transporters are able to confer resistance to tumour cells by removing cytotoxic drugs from intracellular target sites. Clinically these actions provide serious obstacles to effective anticancer chemotherapy. However, these same transporters exist in normal tissues and there provide protection to cells against cytotoxic stresses. Work in the laboratory has been concerned with the mechanisms by which these transporters bring about drug movements [8, 12], looking at some of the agents that impair [5, 10, 13, 14, 15] or can by-pass their actions [16] [Fig 1 shows fluorescent images of doxorubicin inside resistant lung tumour cells following exposure to either a) the free drug or b) its polymer-linked form]. These studies have been undertaken using Hladkyand van Veen), in the Department of Chemical Engineering (Dr M Eccleston) and at the National Cancer Institute, NIH, Bethesda, MA, USA (Dr S Ambudkar)
 |
 |
 |
||
|---|---|---|---|---|
| Fig. 2 Felix RA 2001 PhD, Univ Camb | Figs.1 a & b:Anikina et al 2005 BrJPharmacol online abstract | |||
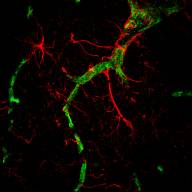
A special focus is now on the localisation [2, 7] and functions of the transporters at the blood-brain barrier where they are known to prevent unwanted substances from entering the brain. [Figure 2 shows a rat brain capillary with P-glycoprotein (stained green) and surrounding astrocytes (stained red)]. Using primary cultures of endothelial cells grown from capillaries that form part of the blood-brain barrier [1, 3, 6, 9, 17] we are exploring the pathways that regulate their expression, seeing how conditions that mimic pathological states ie during toxic, pH, hypoxic and oxidative challenge [6, 9] affect amounts and activities of the various transporters. These studies have been extended to include the influences of other brain cell types, in particular neural precursor cells [18], that in vivo are associated with microvessels in the brain, identifying factors responsible for these cell-cell interactions and the signaling pathways involved. Many aspects of these studies are carried out in close collaboration with Dr S B Hladky whose webpage describes our work on ion transporters. Collaborative links with the Cambridge Centre for Brain Repair and the Department of Neurosurgery at Addenbrooke's Hospital has enabled the studies to be extended from rat to human brain tissue [2, 7].
The laboratory is equipped to undertake tissue isolation, primary cell culture, molecular biological methods for modifying gene expression and for manipulating and analysing RNA, DNA and proteins including real time PCR, Northern, Southern and Western blotting, immunocytochemistry, transport studies with radiolabelled and with fluorescent tracers. Facilities are also available for visualising and quantifying intracellular distribution of fluorescent substrates, analysing intracellular pH and measuring ion channel activity using intracellular recordings.
